SciCheck Digest
The National Institutes of Health has not recommended and the Food and Drug Administration has not approved ivermectin as a COVID-19 treatment. But a Facebook post misleadingly implies that an article published on the NIH website is an endorsement of the drug to treat COVID-19. The NIH and FDA have said more clinical studies are needed.
Full Story
Ivermectin, an antiparasitic medication, has been not approved by the Food and Drug Administration to prevent or treat COVID-19.
 Though researchers have been looking into the drug’s effectiveness for the treatment of COVID-19, the Centers for Disease Control and Prevention, the World Health Organization and the National Institutes of Health have not recommended the use of ivermectin against COVID-19 outside of clinical trials, citing insufficient evidence. (For more, see SciCheck’s “Ongoing Clinical Trials Will Decide Whether (or Not) Ivermectin Is Safe, Effective for COVID-19.”)
Though researchers have been looking into the drug’s effectiveness for the treatment of COVID-19, the Centers for Disease Control and Prevention, the World Health Organization and the National Institutes of Health have not recommended the use of ivermectin against COVID-19 outside of clinical trials, citing insufficient evidence. (For more, see SciCheck’s “Ongoing Clinical Trials Will Decide Whether (or Not) Ivermectin Is Safe, Effective for COVID-19.”)
But a Facebook post misleadingly implies that the NIH endorses a study that comes to a different conclusion about ivermectin than the CDC.
The post juxtaposes a CDC tweet warning against the use of ivermectin with a screenshot of an article on the PubMed website, the National Library of Medicine’s biomedical literature database run by the NIH.
“A tweet from the CDC on ivermectin vs. a study on the NIH website about ivermectin,” reads the caption of the Facebook post from Young Americans for Liberty, a Libertarian organization.
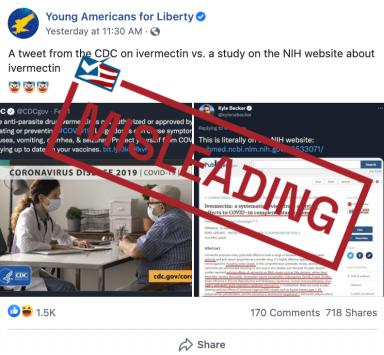
The article, written by two Iranian ophthalmologists, had appeared in The Journal of Antibiotics on June 12, 2020. The article, “Ivermectin: a systematic review from antiviral effects to COVID-19 complementary regimen,” is a review of various studies on the antiviral effects of ivermectin.
The article was posted to PubMed on June 14, 2020. But that doesn’t mean it was endorsed by the NIH, as explained in a disclaimer on the PubMed website:
“The presence of any article, book, or document in these databases does not imply an endorsement of, or concurrence with, the contents by NLM, the National Institutes of Health (NIH), or the U.S. Federal Government.”
In addition, the conclusion of the article concurs with statements released by the CDC and NIH on the need for clinical studies to investigate ivermectin’s safety and effectiveness as a COVID-19 treatment.
The authors of the article, Fatemeh Heidary and Reza Gharebaghi, conclude: “Clinical trials are necessary to appraise the effects of ivermectin on COVID-19 in clinical setting and this warrants additional investigation for probable benefits in humans in the current and future pandemics. … Moreover, further trials are needed to confirm the safety and efficacy of ivermectin for human use against COVID-19 to discover preventive or therapeutic window.”
The CDC tweet pictured in the Facebook post warns against the use of ivermectin to treat COVID-19.
“The anti-parasite drug ivermectin is not authorized or approved by FDA for treating or preventing #COVID19. Large doses can cause symptoms like nausea, vomiting, diarrhea, & seizures,” the CDC tweet reads. “Protect yourself from COVID-19 by staying up to date on your vaccines.” The tweet also links to an FDA update warning against using ivermectin to treat COVID-19.
A statement from the CDC in August said, “Clinical trials and observational studies to evaluate the use of ivermectin to prevent and treat COVID-19 in humans have yielded insufficient evidence for the NIH COVID-19 Treatment Guidelines Panel to recommend its use.”
In its treatment guidelines, the NIH says, “There is insufficient evidence for the COVID-19 Treatment Guidelines Panel (the Panel) to recommend either for or against the use of ivermectin for the treatment of COVID-19. Results from adequately powered, well-designed, and well-conducted clinical trials are needed to provide more specific, evidence-based guidance on the role of ivermectin in the treatment of COVID-19.”
As we’ve written, the CDC recommends the use of ivermectin as a treatment for parasitic infections, such as strongyloidiasis and onchocerciasis, which is otherwise known as “river blindness.”
Editor’s note: SciCheck’s COVID-19/Vaccination Project is made possible by a grant from the Robert Wood Johnson Foundation. The foundation has no control over our editorial decisions, and the views expressed in our articles do not necessarily reflect the views of the foundation. The goal of the project is to increase exposure to accurate information about COVID-19 and vaccines, while decreasing the impact of misinformation.
Sources
CDC (@CDCgov). “The anti-parasite drug ivermectin is not authorized or approved by FDA for treating or preventing #COVID19. Large doses can cause symptoms like nausea, vomiting, diarrhea, & seizures. Protect yourself from COVID-19 by staying up to date on your vaccines.” Twitter. 1 Feb 2022.
Centers for Disease Control and Prevention. “Parasites- Onchocerciasis.” Accessed 7 Feb 2022.
Centers for Disease Control and Prevention. “Parasites – Strongyloides.” Accessed 7 Feb 2022.
Centers for Disease Control and Prevention. “Rapid Increase in Ivermectin Prescriptions and Reports of Severe Illness Associated with Use of Products Containing Ivermectin to Prevent or Treat COVID-19.” CDC.gov. 26 Aug 2021.
Gore, D’Angelo. “Social Media Posts Repeat Inaccurate Reporting on Ivermectin and Omicron.” FactCheck.org. 2 Feb 2022.
Hancock, Tess. “Social Media Post on Use of Ivermectin for Refugees Lacks Context.” FactCheck.org. 17 Sep 2021.
Heidary, Fatemeh and Reza Gharebaghi. “Ivermectin: a systematic review from antiviral effects to COVID-19 complementary regimen.” The Journal of Antibiotics. 12 Jun 2020.
Heidary, Fatemeh and Reza Gharebaghi. “Ivermectin: a systematic review from antiviral effects to COVID-19 complementary regimen.” PubMed. 17 May 2020.
Jaramillo, Catalina. “Ongoing Clinical Trials Will Decide Whether (or Not) Ivermectin Is Safe, Effective for COVID-19.” FactCheck.org. Updated 29 Oct 2021.
Clinical Trials.gov. National Institutes of Health. Accessed 7 Feb 2022.
U.S. Food and Drug Administration. “Why You Should Not Use Ivermectin to Treat or Prevent COVID-19.” Fda.gov. 10 Dec 2021.
World Health Organization. “WHO advises that ivermectin only be used to treat COVID-19 within clinical trials.” WHO.int. 31 Mar 2021.
National Institute of Health. “COVID-19 Treatment Guidelines Ivermectin” NIH.gov. Updated 11 Feb 2021.
PubMed Central. “Disclaimer.” Ncbi.nlm.nih.gov. Accessed 7 Feb 2022.