Social media posts are misinterpreting the results of a Centers for Disease Control and Prevention report, which found 74% of people in a COVID-19 outbreak were vaccinated, to argue against immunization. But experts say the statistic is misleading without more context — and doesn’t mean that the vaccines don’t work.
Misconception: Vaccination
COVID-19 Surges Among Unvaccinated in Florida, Contrary to Baseless Claims
Florida health officials have reported a 60% rise in COVID-19 cases, and hospitals are reporting that 95% of COVID-19 patients are not fully vaccinated. But a Facebook post makes the baseless claims that Florida’s numbers are not going up, and that all COVID-19 patients recently admitted to a Palm Beach County hospital had been vaccinated.
Vaccines Remain Largely Effective Against Delta Variant, Counter to Claims From Fox News Guest
Multiple studies show the FDA-authorized COVID-19 vaccines continue to be effective against the delta variant of the coronavirus, even if the potency of the vaccines is somewhat reduced. But a guest on Fox News falsely claimed the delta variant “really is not responsive at all, or protected at all by the vaccines” and there is “no clinical reason to go get vaccinated.”
Posts Baselessly Link COVID-19 Tests to Vaccine Conspiracy Theory
The COVID-19 vaccines currently in use must be administered via injection. But Instagram posts baselessly suggest that Bill Gates and George Soros will use COVID-19 tests to secretly vaccinate people who haven’t yet received the shots. There is no evidence for that conspiracy theory, and scientists say trying to administer a vaccine with a swab would likely not be effective.
Video Twists Advice on Delta Variant and Vaccination
An epidemiologist recommended that people get the COVID-19 vaccine because some evidence suggests an unvaccinated person who gets the delta variant is “twice as likely to require hospital treatment” than someone infected with the alpha variant. But a Facebook video twists that advice to claim that he said vaccinated people would be twice as likely to be hospitalized.
Viral Posts Lift Bogus ‘Quarantine’ Story from Satire Site
Mayim Bialik and Sons Got COVID-19 Vaccine
Photo Shows 2018 France World Cup Celebration, Not Vaccine Protest
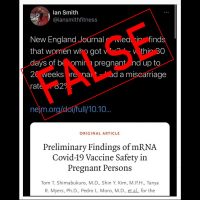
CDC Data Thus Far Show COVID-19 Vaccination Safe During Pregnancy
Federal vaccine monitoring systems have identified no safety concerns with the COVID-19 vaccines for pregnant people. Preliminary Centers for Disease Control and Prevention data show that miscarriage is not more frequent than expected in vaccinated people. Online posts, however, falsely contend that such data, as reported in a CDC publication, show an 82% miscarriage rate.